LTS provides alternative drug delivery systems in the form of transdermal therapeutic systems (TTS) and oral thin films (OTF).
At LTS we manufacture for global markets of your choice, making use of our expertise as an innovative market leader.
LTS develops and manufactures drug delivery systems that offer patients reliable therapeutic efficacy with total comfort and convenience.
LTS provides alternative drug delivery systems in the form of transdermal therapeutic systems (TTS), oral thin films (OTF) and micro array patches (MAP).
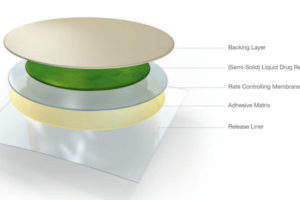
TTS are applied directly to the skin. The active substance is absorbed by the skin and distributed through the body via the bloodstream. This innovative mode of administration has improved the simplicity of treatment for patients for almost 30 years.

Oral thin films are loaded with active substances. The Thin films are taken orally and dissolve immediately in the mouth or are applied to the mucosa. For transmucosal films, the active substance enters the bloodstream directly via the oral mucosa, without having to first pass through the gastrointestinal tract.

LTS’ research and development (R&D) focuses on new technologies to complement and expand the potential of alternative drug delivery systems. It is continually developing its TTSs and OTFs, but the newest technology is the Micro Array Patch (MAP). The future of drug delivery. It helps to enhance the delivery of a drug through the transdermal/intradermal route overcoming various problems associated with the conventional formulations. It is based on a dissolvable microneedle technique, and it offers many advantages for a custom-made design. Advantages are

As a business-to-business (B2B) partner LTS does not sell any of its products, making it a dedicated technology partner to the pharmaceutical industry, rather than a competitor against classical pharmaceutical companies. In addition, LTS does not develop any active substances. Instead, it focuses its expertise on the development of new, innovative drug delivery systems that specialize in the transdermal, transmucosal and intradermal application of existing and novel agents.
Based on a successful feasibility study, LTS develops the ideal delivery system for each customer and each substance.
Patients aren’t just numbers to us, they are people. That’s why LTS is unrelenting in its individual and collective commitment to make life better. For our partners. For their patients. For the LTS family.
LTS Combines its market-leading expertise, experience and enterprise, and works with its partners to create patient-centred technologies.
From bench to market, LTS delivers value that exceeds expectations and remains the trusted partner in innovative oral and transdermal drug delivery systems.
To achieve success in TTS and OTF, you need a strong, experienced and committed pharmaceutical partner at your side. As a market leader, LTS fulfils all these requirements.
LTS continues to invest in flexible, state of the art production equipment that supports its partners needs in terms of high precision, high quality, bespoke products.
LTS has the experience and expertise to develop and manufacture successful products right up to market approval. It has sufficient capacity to manufacture the batch sizes required for global sales and high-volume products. With a 60,000m² manufacturing footprint LTS produces almost a billion systems every year and features in excess of 40 conversion lines, 100 mixing vessels, more than ten coating lines for different technologies and multiple packaging/serialization lines.

Two identical production facilities in Germany and the US secure supply chain reliability even in crisis periods.
LTS is recognized by all major health authorities worldwide and has broad experience in the field of hormone therapy and anaesthetics narcotic substances. It systematically separates the production of hormone products from non-hormone products to avoid any risk of possible cross-contamination.
LTS supports each client with a development team that can be integrated into the clients’ research teams. LTS provides customers with total transparency in all project phases.
LTS Lohmann Therapie-Systeme AG
Lohmannstraße 2
D-56626 Andernach
Germany
Phone: (0 26 32) 99-0
Fax: (0 26 32) 99 22 00
Email: info@ltslohmann.com
Website: ltslohmann.com