The US Food and Drug Administration (FDA) has finished a filing review of Valneva’s Biologics License Application (BLA) for single-shot chikungunya vaccine candidate VLA1553.
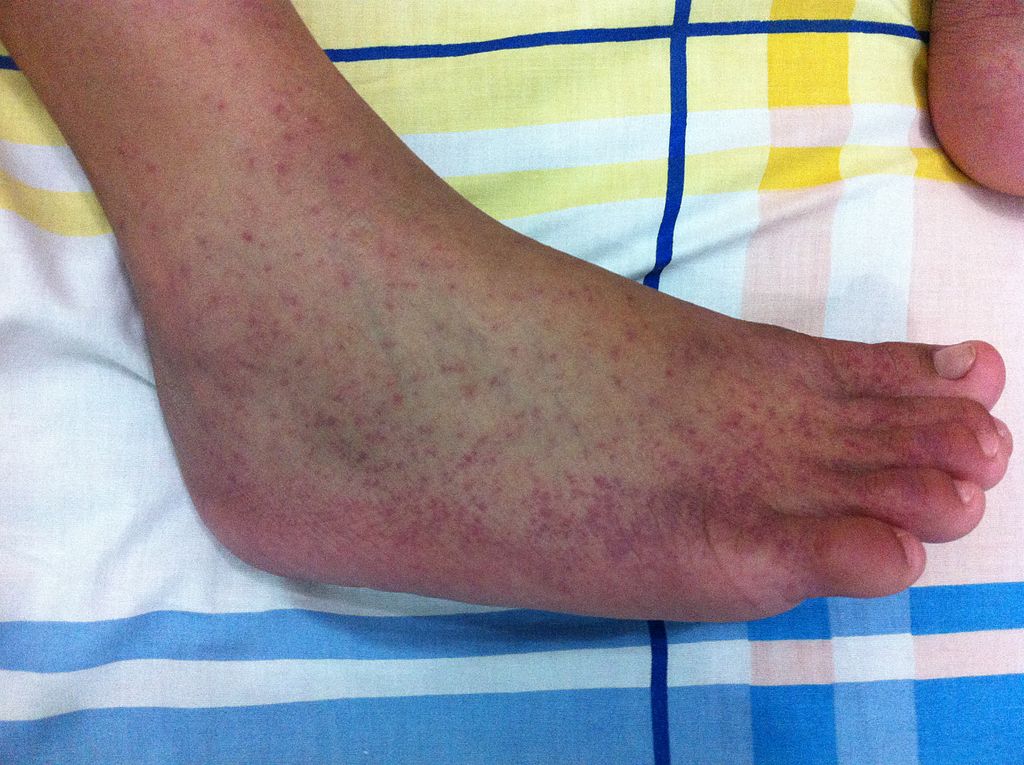
Image: A mosquito-borne viral disease, Chikungunya is caused by the chikungunya virus. Credit: Nsaa/ commons.wikimedia.org.
Subscribe to our email newsletter
The regulatory agency has ascertained that the application is sufficiently finished to allow a substantive review.
Valneva, a specialty vaccine company, stated that the review is classified as priority.
VLA1553 has been assigned a Prescription Drug User Fee Act (PDUFA) review goal date at the end of August this year.
By this date, the US FDA plans to take action on the application.
The FDA’s acknowledgement of filing, however, does not imply that a license will be granted, nor does it represent any analysis of the sufficiency of the data submitted.
A live-attenuated, single dose investigational vaccine candidate, VLA1553 targets the chikungunya virus, which has spread to more than 100 countries.
This vaccine has been designed by removing a part of the chikungunya virus genome.
A mosquito-borne viral disease, Chikungunya is caused by the chikungunya virus (CHIKV), a Togaviridae virus, transmitted by Aedes mosquitoes.
In January 2021, Valneva and Instituto Butantan in Brazil signed an agreement for the development, manufacturing and marketing of VLA1553 in order to make the caccine more accessible to low- and middle-income countries (LMIC).
In March 2022 Valneva reported final data from the pivotal Phase 3 trial of VLA1553, final lot-to-lot consistency results in May 2022 as well as positive twelve-month persistence data in December 2022.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.