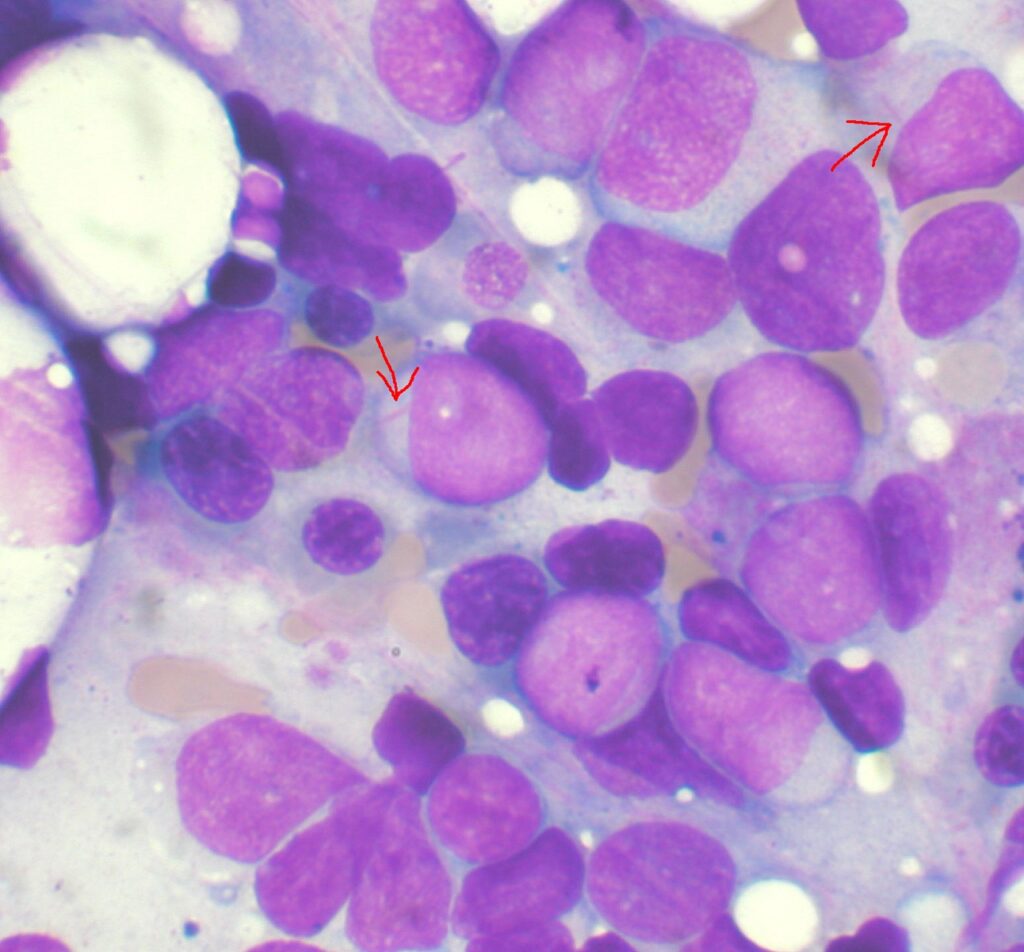
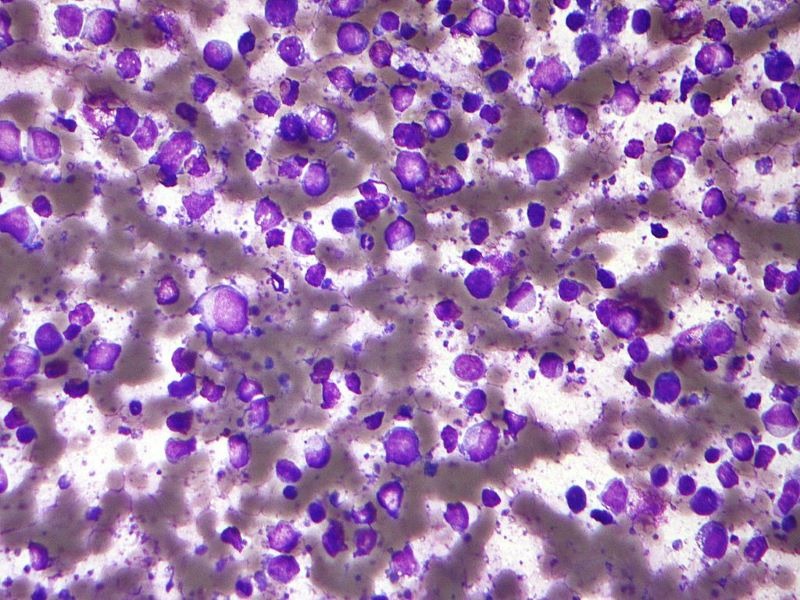
FDA grants priority review for Daiichi Sankyo’s AML medicine
The regulator has accepted the company’ new drug application (NDA) for the combination therapy and as continuation of quizartinib monotherapy after consolidation to treat FLT3-ITD–positive AML patients. Quizartinib,
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.