The US Food and Drug Administration (FDA) has accepted Merck’s (MSD) biologics license application (BLA) of sotatercept to treat pulmonary arterial hypertension [PAH (WHO Group 1)], for priority review.
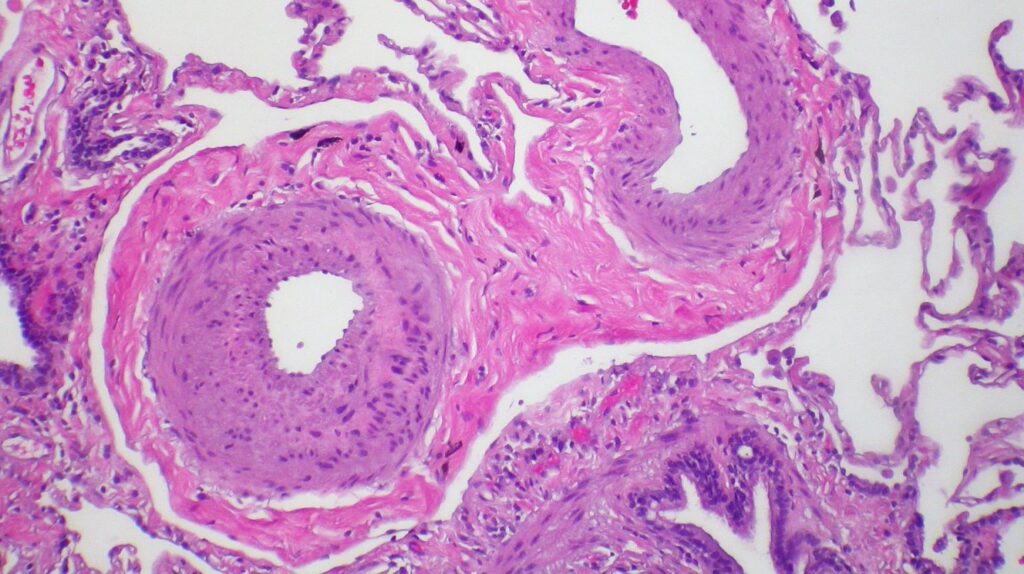
Merck filed BLA for sotatercept based on findings from the Phase III STELLAR clinical trial in pulmonary arterial hypertension patients. Credit: Yale Rosen from USA / commons.wikimedia.org.
Subscribe to our email newsletter
An investigational activin signalling inhibitor, sotatercept is intended for usage in adult PAH patients.
A regulatory decision on the approval of the therapy is anticipated on 26 March 2024.
The company filed BLA for sotatercept based on findings from the Phase III STELLAR clinical trial.
In the trial, sotatercept plus background therapy showed to provide improvement in six-minute walk distance (6MWD) as well as eight of the nine secondary outcomes.
On receipt of FDA approval, sotatercept will become a first-of-its-kind approach to manage a rare ailment of the pulmonary arteries.
Merck Research Laboratories global clinical development senior vice-president Dr Joerg Koglin said: “Despite advances in the treatment of PAH over the last two decades, there is still a significant need to improve outcomes for patients.
“The FDA’s acceptance of this application is an exciting milestone in our journey to bring this novel activin signalling inhibitor to patients.
“Based on the profound improvements across primary and secondary outcome measures in the Phase III STELLAR trial, we believe sotatercept has the potential to transform the treatment of patients with PAH.”
The company noted that sotatercept was demonstrated to reduce vascular cell proliferation thereby reversing the remodelling of the vascular and right ventricle in pre-clinical research.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.

