Aurinia Pharmaceuticals’ collaboration partner Otsuka Pharmaceutical has submitted a new drug application (NDA) to the Japanese Ministry of Health, Labour and Welfare for voclosporin to treat lupus nephritis (LN).
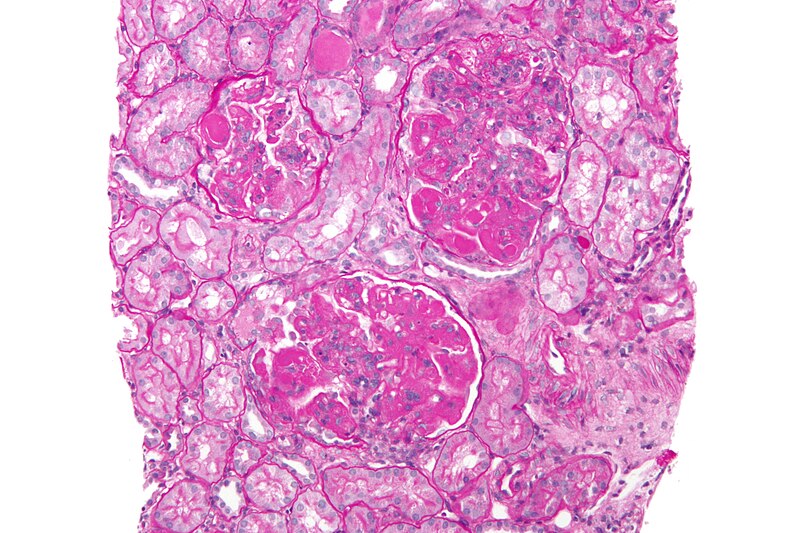
High magnification micrograph of diffuse proliferative lupus nephritis, class IV. Credit: Nephron / commons.wikimedia.org.
Subscribe to our email newsletter
The company filed the application to manufacture and sell the second-generation oral calcineurin inhibitor, voclosporin, in Japan.
In January 2021, voclosporin along with a background immunosuppressive therapy regimen received the US Food and Drug Administration (FDA) approval to treat adults with active LN.
At present, it is available in the country under the LUPKYNIS brand name.
LUPKYNIS is the first US FDA- and EC-approved oral medicine intended to treat adults with active LN and is a structurally modified calcineurin inhibitor with a dual mechanism of action.
Aurinia and Otsuka signed a collaboration and licensing agreement in December 2020 to develop and commercialise voclosporin in the EU, Japan, the UK, Russia, Switzerland, Norway, Belarus, Iceland, Liechtenstein, and Ukraine to treat LN.
Aurinia Pharmaceuticals president and CEO Peter Greenleaf said: “Lupus nephritis is a serious, complex condition associated with SLE, that can benefit from advanced therapies that focus toward the treatment goal of kidney preservation.
“We look forward to continuing to work with Otsuka to expand availability to voclosporin and provide patients and physicians access to this new therapy that addresses a significant unmet need in lupus nephritis.”
LN is a complication of the autoimmune disease systemic lupus erythematosus (SLE) that impacts the kidneys seriously.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.

