Iovance Biotherapeutics has completed submission of the Biologics License Application (BLA) for lifileucel to the US Food and Drug Administration (FDA).
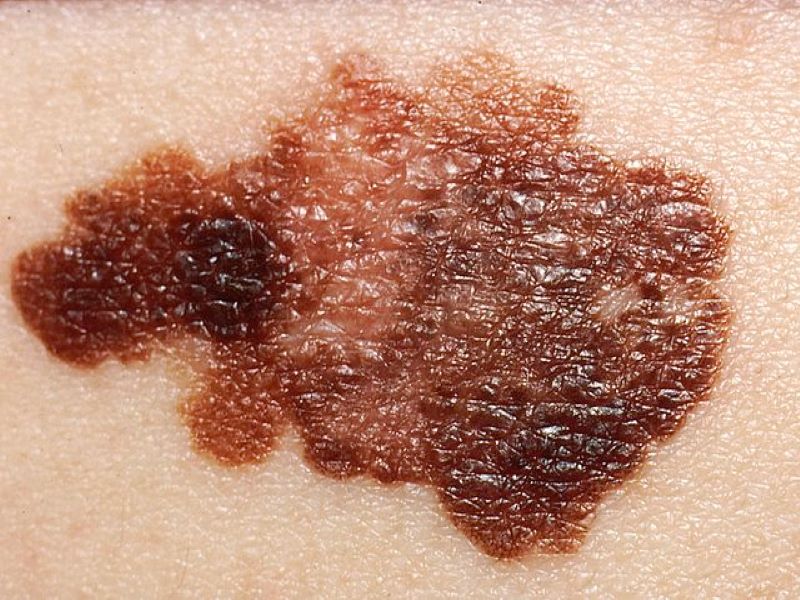
The slide shows melanoma on a patient's skin. Credit: Doc James / commons.wikimedia.org.
Subscribe to our email newsletter
A tumour infiltrating lymphocyte (TIL) therapy, lifileucel is designed to treat patients with advanced (unresectable or metastatic) melanoma who advanced on or after prior anti-PD-1/L1 therapy and targeted therapy.
Iovance has submitted BLA based on positive clinical data from the C-144-01 clinical study in patients with advanced post-anti-PD1 melanoma.
Furthermore, the company signed an agreement with the FDA for the registrational trial design related to the Phase 3 TILVANCE-301 trial of lifileucel along with pembrolizumab in patients with frontline advanced melanoma.
This study is expected to support complete approval of lifileucel in post-anti-PD-1 advanced melanoma besides supporting registration for lifileucel along with pembrolizumab as therapy to treat advanced melanoma in the frontline setting.
Startup activities related to this trial are ongoing. The study is anticipated to be underway at the time of potential accelerated approval for the therapy.
The regulator has 60 days to determine the acceptability of the BLA for assessment after receipt of the BLA submission for lifileucel.
Iovance interim president and CEO Frederick Vogt said: “Completing our BLA submission for lifileucel is a critical step forward in our journey to deliver the first individualised, one-time cell therapy for a solid tumour.
“I would like to acknowledge the patients and physicians who participated in the C-144-01 clinical trial and the FDA review team for their commitment and support, as well as our internal team for their tremendous effort in completing the first BLA submission for Iovance.”
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.

