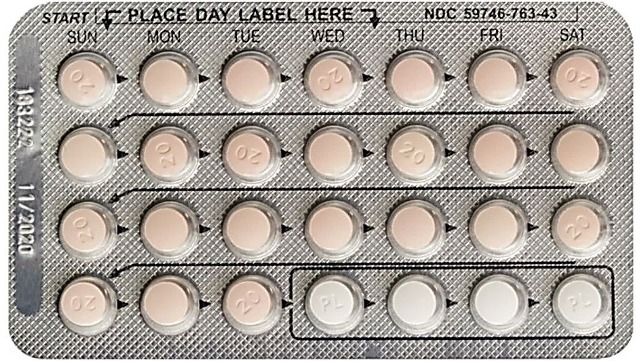
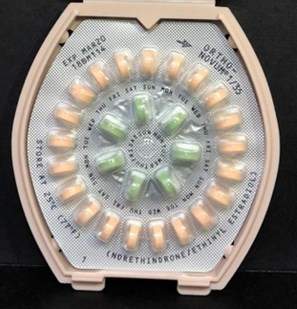
Jubilant recalls one lot of Drospirenone and Ethinyl Estradiol tablets
Jubilant Cadista Pharmaceuticals is voluntarily recalling one lot of Drospirenone and Ethinyl Estradiol Tablets, USP, 3 mg/ 0.02mg, 28x3 Blister Pack/Carton to the consumer level.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.