A clinical stage biotechnology company Immunomic Therapeutics has received Fast Track Designation (FTD) from the US Food and Drug Administration (FDA) for its ITI-3000 programme to treat patients with Merkel Cell Carcinoma (MCC).
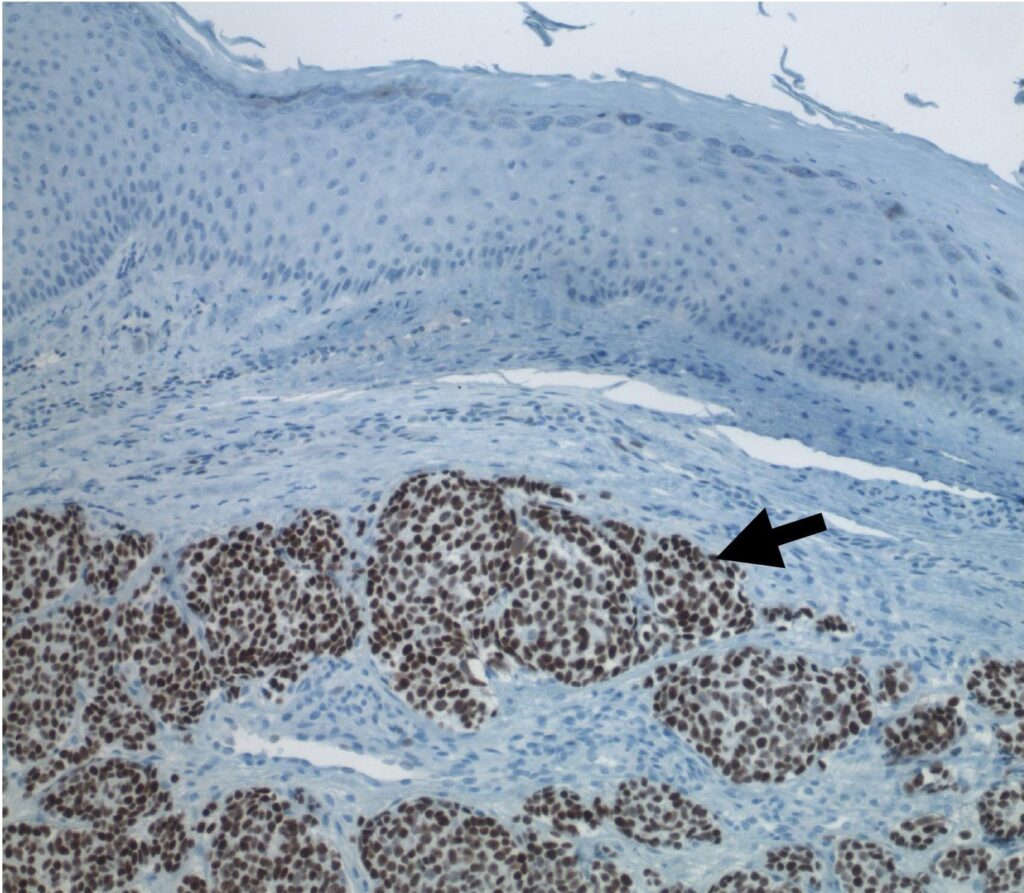
Image: MCC is a rare but aggressive form of skin cancer typically caused by Merkel cell polyomavirus (MCPyV). Credit: Patrick S. Moore via commons.wikimedia.org.
Subscribe to our email newsletter
Currently, the company is enrolling a Phase 1 trial analysing ITI-3000, a plasmid DNA (pDNA) vaccine targeting patients with MCC.
MCC is a rare but aggressive form of skin cancer typically caused by the Merkel cell polyomavirus (MCPyV).
Immunomic Therapeutics CEO Dr. William Hearl said: “The FDA’s decision to grant FTD underscores the potential for the ITI-3000 program to address a serious unmet need and serve as a meaningful therapeutic option for patients with Merkel cell carcinoma.
“We are committed to unlocking the full potential of ITI-3000 in patients with this aggressive form of skin cancer. We expect to report top-line data from our ongoing phase 1 trial of ITI-3000 in MCC patients next year and look forward to working closely with the FDA on a potential next phase clinical study design, while simultaneously continuing dialogue with possible partners.”
ITI-3000 programme leverages Immunomic Therapeutics’ investigational UNiversal Intracellular Targeted Expression (UNITE) platform, which is powered by LAMP (Lysosome Associated Membrane Protein) that fuses sequences from the mutated form of MCPyV’s large T antigen (LT) into the LAMP-1 gene sequence.
This lysosomal targeting technology has shown to lead to enhanced antigen presentation and a balanced immune response, including, ITI-3000 activated antigen-specific CD4+ T cells in vivo.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.