Genkyotex’s liver disease drug GKT831 succeeds in phase 2 trial
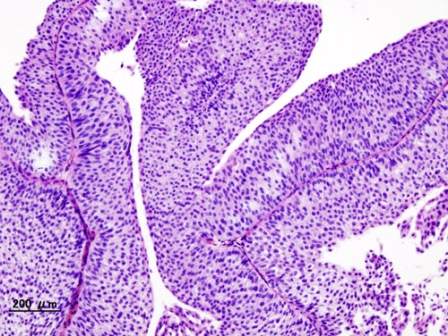
Genkyotex’s NOX1/4 inhibitor GKT831 has succeeded in a phase 2 trial in primary biliary cholangitis (PBC), a type of liver disease, by meeting both primary and secondary interim efficacy endpoints.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.