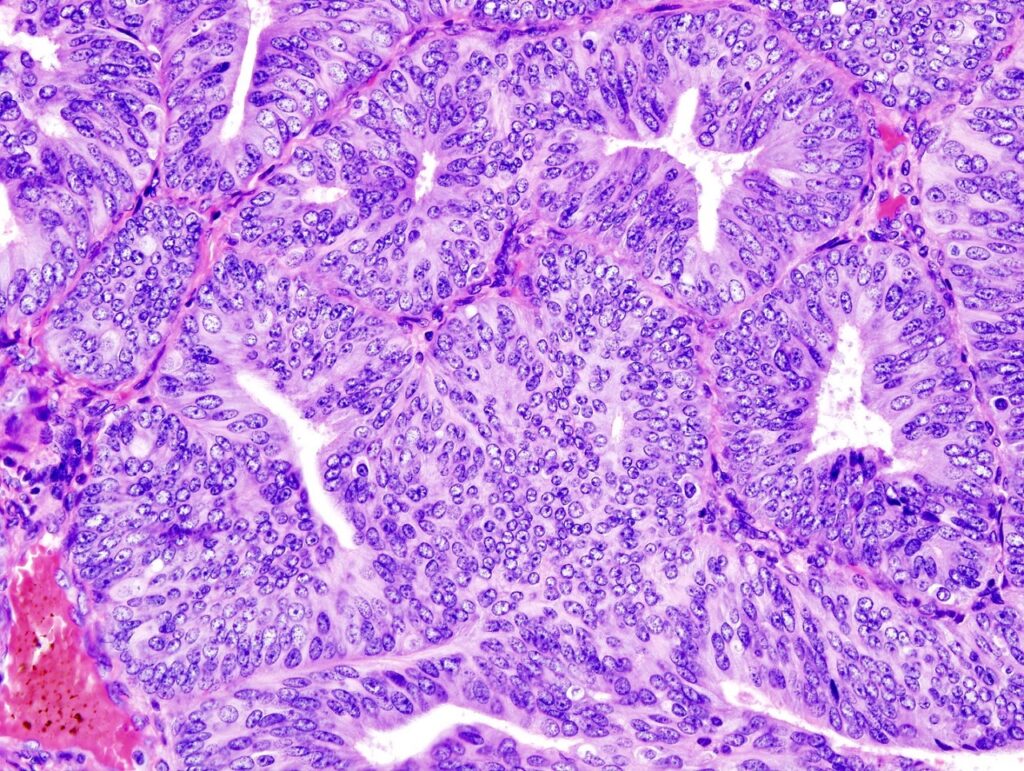
FDA approves Merck, Eisai’s Keytruda and Lenvima combination for endometrial cancer
Merck and Eisai have secured approval from the US Food and Drug Administration (FDA) for Keytruda (pembrolizumab) plus Lenvima (lenvatinib) combination to treat patients with certain types of advanced endometrial carcinoma.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.