Pfanstiehl provides parenteral-grade excipients and highly potent active pharmaceutical ingredients (API) in accordance with current good manufacturing practice (cGMP) guidelines.
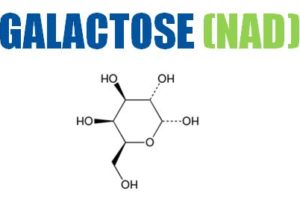
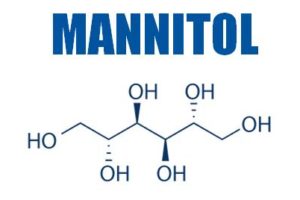
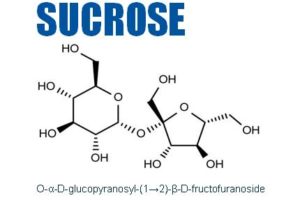
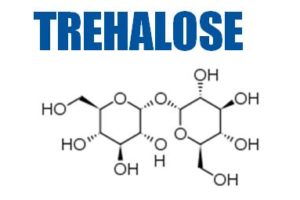
The company develops high-purity, low-endotoxin (HPLE) carbohydrates such as trehalose, sucrose, mannitol, galactose, maltose and mannose for use as injectable excipients for the stabilisation of proteins, monoclonal antibodies (mAb) and vaccines.
Pfanstiehl’s HPLEs are used as supplements for industrial cell culture, cell therapy and cryopreservation media.
Pfanstiehl works closely with many of the world’s largest multinational pharmaceutical and biopharmaceutical firms, as well as virtual pharmaceutical companies.
It synthesises proprietary and commercial compounds in both large and small quantities.
Pfanstiehl has a 40-year history in custom synthesis, manufacturing and process scale-up development.
Pfanstiehl provides high-quality plug-and-play solutions to biosimilar and pharmaceutical firms worldwide. It provides custom synthesis of new chemical entities (NCE), speciality carbohydrates and injectable-quality, cGMP pharmaceutical ingredients in commercial quantities.
The company’s capabilities include process scale-up, development and process validation, according to cGMP guidelines. Its facilities and equipment are configured to produce HPLE compounds, including potent (highly active toxic or cytotoxic) drugs under cGMP conditions.
As a long-time manufacturer of low-endotoxin carbohydrates, injectable-grade excipients, active pharmaceutical ingredients (API) and pharmaceutical intermediates, Pfanstiehl can establish and maintain US and European drug master files (DMF) and European Certificates of Suitability. Pfanstiehl helps you get your compound to market, bridging the gap between product development and regulatory approval.
Pfanstiehl performs a wide range of chemistries to meet client requirements, including:
Pfanstiehl’s purification offerings include extraction and isolation, selective crystallisation and ultrafiltration / nanofiltration, as well as deionisation, electrodialysis, activated carbon polishing and chiral resolution.
With more than 40 years’ cGMP manufacturing experience, Pfanstiehl has prepared the necessary documentation, validation and qualification systems to ensure compliance with the FDA and other regulatory bodies worldwide.
Maintaining more than 15 DMFs for customers and for Pfanstiehl proprietary APIs, the company’s services include cGMP manufacturing and cGMP-compliant process development and validation.
Pfanstiehl’s facilities are FDA registered and inspected with more than 15 active DMFs, which include US Type II, Canadian and European DMFs and Certificates of Suitability. Fully qualified equipment and utilities are used in the cGMP manufacturing areas, in addition to cGMP development and scale-up services.
Experienced manufacturing, quality assurance and regulatory affairs teams are available to provide support and advice during the devlopment process.
The analytical services laboratory is equipped with modern analytical testing instrumentation, which includes:
The company’s process chemistry lab has the following analytical instruments and lab equipment, including:
Pfanstiehl, Inc.
1219 Glen Rock Avenue
Waukegan, IL
60085-0439
United States of America
Tel: +1 847 623 0370
Fax: +1 847 623 9173
Email: cs@pfanstiehl.com
www.pfanstiehl.com/api