The UK’s Medicines and Healthcare Products Regulatory Agency (MHRA) has granted Alnylam Pharmaceuticals’ patisiran, an investigational RNAi therapeutic for the treatment of hATTR amyloidosis, a positive scientific opinion through the Early Access to Medicines Scheme (EAMS).
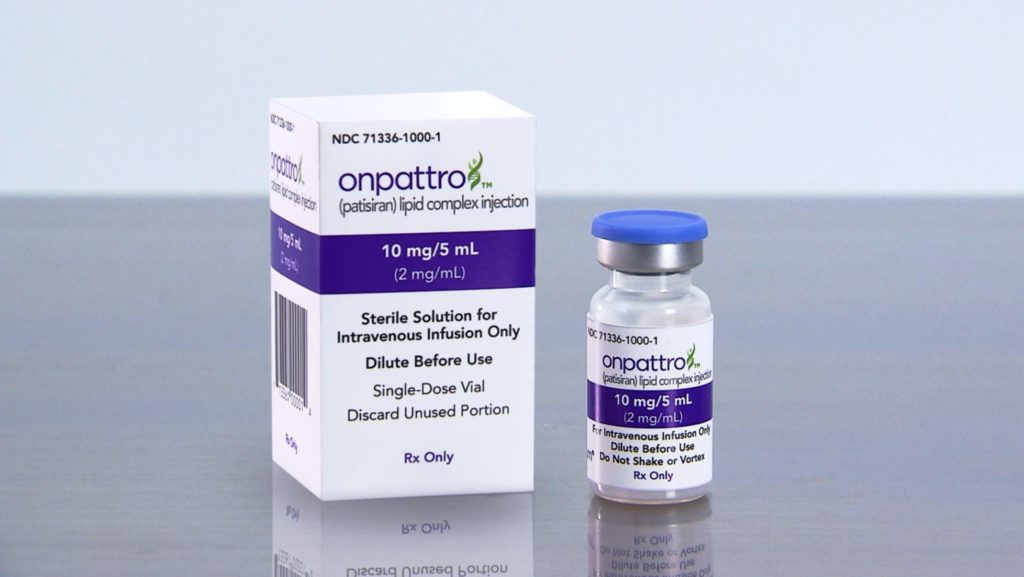
Image: Alnylam’s Onpattro packaging and product vial. Photo: courtesy of Business Wire.
Subscribe to our email newsletter
With this decision, eligible adults in the UK with hATTR amyloidosis can gain access to patisiran before the drug is granted marketing authorization by the European Commission (EC).
The aim of EAMS is to provide early availability of innovative, new, unlicensed medicines to UK patients who have a high degree of unmet clinical need. The medicines included in the scheme are those that are intended to treat, diagnose or prevent seriously debilitating or life-threatening conditions where there are no adequate treatment options.
Alnylam UK & Ireland country manager Brendan Martin said: “The EAMS positive scientific opinion reflects Alnylam’s commitment to patients with hATTR amyloidosis and their families, for whom it will be welcome news.
“New treatment options that impact the underlying cause of the disease, improve neuropathy and a patient’s ability to function on a daily basis, are urgently needed and this decision will allow patients to have access to patisiran without delay.”
The MHRA’s decision is based on the evaluation of the effects of patisiran in hATTR amyloidosis patients with polyneuropathy and its safety profile as demonstrated in the APOLLO Phase 3 study.
The results of the APOLLO study were published July 5, 2018 in The New England Journal of Medicine (NEJM). Within EAMS, patisiran will be made available for eligible hATTR amyloidosis patients presenting with symptoms of polyneuropathy and/or cardiomyopathy.
MHRA’s decision follows the recent positive opinion by the CHMP for patisiran for the treatment of hATTR amyloidosis in adult patients with stage 1 or stage 2 polyneuropathy, with the EC decision expected in September.
If approved by the EC, the medicine will be commercialized under the brand name ONPATTRO.
Patisiran is currently under priority review as a Breakthrough Therapy with the U.S. Food and Drug Administration (FDA), with an action date of August 11, 2018. Regulatory filings in other markets, including Japan, are planned starting in mid-2018.
Source: Company Press Release
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.