Danish medical dermatology firm Leo Pharma announced that its tralokinumab has achieved all primary and secondary endpoints in three phase 3 studies for the treatment of adults with moderate-to-severe atopic dermatitis (AD).
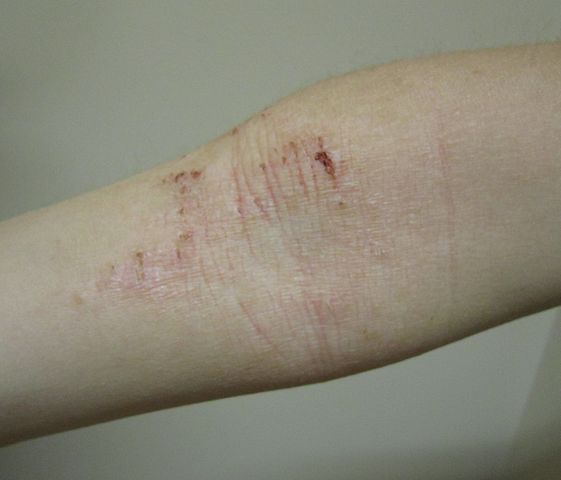
Image: Atopic dermatitis of the inside crease of the elbow. Photo: courtesy of James Heilman, MD.
Subscribe to our email newsletter
The overall adverse event rate was comparable between tralokinumab and placebo during the trials, said the company.
Tralokinumab is an investigational and fully human monoclonal antibody, which neutralises the interleukin-13 (IL-13) cytokine, a crucial driver of the type 2 inflammation that plays a significant role in AD.
Atopic dermatitis is a chronic, relapsing skin disease, which is identified by severe itch, dry skin, persistent immune-mediated inflammation and skin barrier defects.
ECZTRA 1 and ECZTRA 2 randomised, double-blind, placebo-controlled, multinational and 52-week studies enrolled 802 and 794 adult patients to assess the efficacy and safety of tralokinumab as monotherapy in adults with moderate-to-severe AD who are candidates for systemic therapy.
ECZTRA 3 is a randomised, double-blind, placebo-controlled and multinational 32-week study that recruited 380 adult patients to assess the efficacy and safety of tralokinumab in combination with topical corticosteroids (TCS) in patients with moderate-to-severe AD who are candidates for systemic therapy.
An Investigator Global Assessment (IGA) score of clear (0) or almost clear (1) skin at week 16 and at least a 75% or greater change from baseline in their Eczema Area and Severity Index (EASI) score at week 16 are the primary endpoints in the three studies.
The secondary endpoints include a change from baseline to week 16 in Scoring of atopic dermatitis (SCORAD), Pruritus Numeric Rating Scale (NRS) of at least four, and Dermatology Life Quality Index (DLQI).
The company intends to submit marketing authorisation applications for tralokinumab to treat adult patients with moderate-to-severe AD to regulatory agencies in 2020.
Leo Pharma global research and development executive vice president Dr Kim Kjoeller said: “Despite recent treatment advances, we consistently hear from healthcare professionals around the world that additional treatment options are needed to address the different signs and symptoms for each patient.
“We are encouraged by these study results, which show that tralokinumab could be an efficacious and well-tolerated long-term treatment solution for patients living with this debilitating chronic skin disease.”
In August 2018, Leo Pharma agreed to acquire the global prescription dermatology business of Bayer for an undisclosed price.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.

