Janssen Pharmaceutical, part of Johnson & Johnson, has secured marketing authorisation from the European Commission (EC) for its Ebola vaccine regimen to prevent Ebola virus disease.
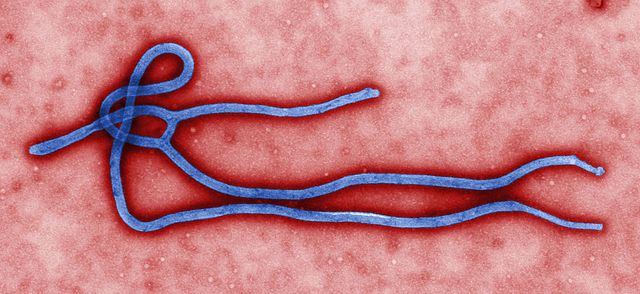
Electron micrograph of an Ebola virus virion. (Credit: CDC/Cynthia Goldsmith)
Subscribe to our email newsletter
As a result of the approval, Janssen is currently collaborating with the World Health Organisation (WHO) on vaccine pre-qualification to advance the registration of its preventive Ebola vaccine regimen in African countries.
Janssen’s Ebola vaccine regimen is specified for active immunisation to prevent Ebola virus disease caused by the Zaire ebolavirus species in individuals aged one year and above.
Janssen Research & Development global head Dr Mathai Mammen said: “The approval of our Ebola vaccine symbolizes the progress Janssen has made towards achieving our vision of delivering potentially transformational vaccines to communities most at risk of deadly infectious diseases.”
The company has submitted two marketing authorisation applications (MAAs) to the European Medicines Agency (EMA) for the vaccines including a two-dose regimen such as Zabdeno (Ad26.ZEBOV) and Mvabea (MVA-BN-Filo).
The regimen is comprised of Ad26.ZEBOV as the first dose, which is based on Janssen’s AdVac viral vector technology. The MVA-BN-Filo is the second dose, which is based on Bavarian Nordic’s MVA-BN technology and administered approximately eight weeks later.
According to the company, the danger Ebola outbreak to date was the West African epidemic, which resulted in around 30,000 cases and over 11,000 deaths between 2014 and 2016.
Janssen’s AdVac technology is also being used for the development of a vaccine candidate to prevent Covid-19 disease.
Johnson & Johnson chief scientific officer and executive committee vice-chairman Dr Paul Stoffels said: “The European approval of Janssen’s Ebola vaccine regimen is a landmark moment – both for our Company and in the world’s battle against the deadly Ebola virus. Building on our history, we are committed to bringing forward vaccines to help overcome the threat of some of the world’s most life-threatening infectious diseases.”
In June this year, Janssen Pharmaceutical secured EC approval for the subcutaneous formulation of Darzalex (daratumumab) for the treatment of multiple myeloma in adult patients.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.

