Health Canada has granted approval for VBI Vaccines’ PreHevbrio [3-antigen Hepatitis B Vaccine (Recombinant)] to prevent Hepatitis B virus (HBV) in adults.
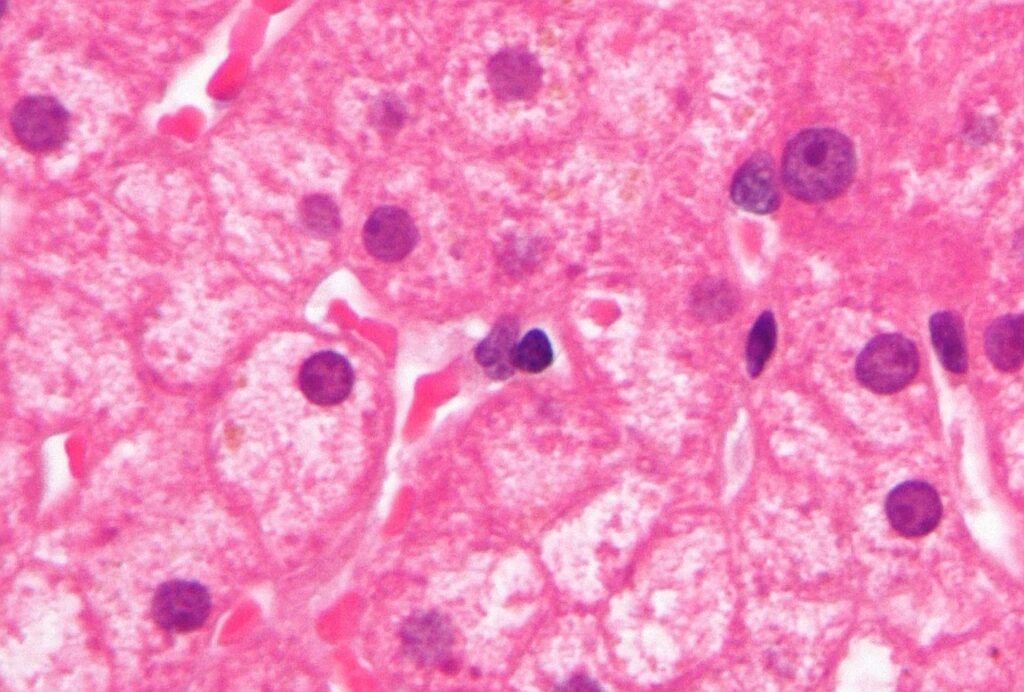
Micrograph of ground glass hepatocytes, as seen in a chronic hepatitis B infection. Credit: Nephron / commons.wikimedia.org.
Subscribe to our email newsletter
PreHevbrio has been approved for active immunisation against infection which is caused by all known subtypes of HBV in people aged 18 years and above.
It is said to be the only 3-antigen hepatitis B vaccine that includes three hepatitis B surface antigens, S, pre-S1, and pre-S2, of the HBV.
The vaccine is already approved for use in the US, the UK, Israel, Canada, and the European Union/European Economic Area.
VBI Vaccines president and CEO Jeff Baxter said: “We are excited to announce Health Canada’s approval, a fourth regulatory approval for this vaccine, and an achievement that is another meaningful step in our effort to provide broad access to our 3-antigen hepatitis B vaccine.
“As we’ve said many times, we believe PreHevbrio has the potential to be a meaningful and differentiated tool that can help healthcare providers make a difference in the fight to eradicate hepatitis B, and we look forward to supporting public health initiatives in Canada to facilitate this.”
The latest regulatory approval was based on data obtained from two pivotal, double-blind, randomised, controlled Phase III clinical trials, PROTECT and CONSTANT.
The two trials compared PreHevbrio to a single-antigen HBV vaccine, Engerix-B.
In the PROTECT study, data demonstrated that the participants treated with PreHevbrio had significant higher rates of seroprotection compared to the Engerix-B.
Myalgia, fatigue, injection site pain and tenderness are the most common adverse events observed in all age groups.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.

