Celltrion Healthcare gets EC approval for its subcutaneous formulation of biosimilar infliximab, Remsima
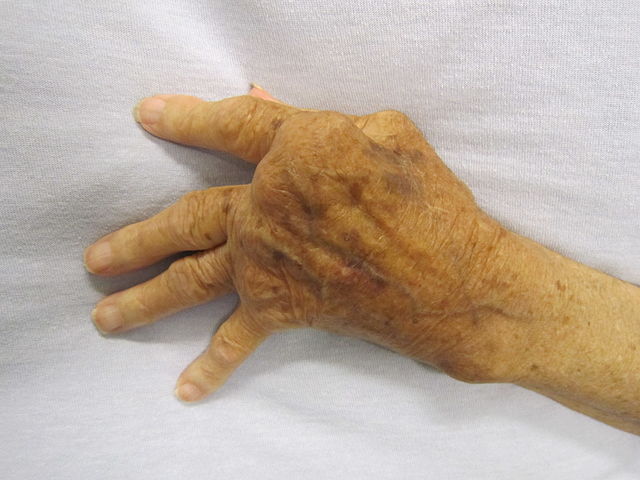
Celltrion Healthcare has secured approval from the European Commission (EC) for its Remsima SC (CT-P13 SC), a subcutaneous formulation of biosimilar infliximab, Remsima, to treat patients with rheumatoid arthritis (RA).
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.