Shionogi announced FETROJA (cefiderocol) is now available in the U.S. for patients 18 years of age or older who have limited or no alternative treatment options, for the treatment of complicated urinary tract infections (cUTIs), including pyelonephritis, caused by the following susceptible Gram-negative microorganisms: Escherichia coli (E. coli), Klebsiella pneumoniae (K. pneumoniae), Proteus mirabilis (P. mirabilis), Pseudomonas aeruginosa (P. aeruginosa), and Enterobacter cloacae (E. cloacae) complex.
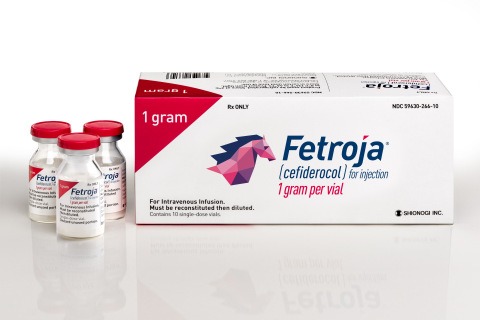
FETROJA is now available in the U.S. for patients 18 years of age or older who have limited or no alternative treatment options for complicated urinary tract infections (Credit: Shionogi Inc)
Subscribe to our email newsletter
FETROJA, which was approved by the U.S. Food and Drug Administration on November 14, 2019, is the first approved antibiotic that functions as a siderophore and has the ability to overcome many of the resistance mechanisms that Gram-negative bacteria employ against antibiotics.
Under FETROJA’s Qualified Infectious Disease Product (QIDP) status, approval of the cUTI indication was based on limited clinical safety and efficacy data.
“The infectious disease community faces a serious challenge in combating cUTIs caused by Gram-negative pathogens, leading to high mortality rates. This illustrates a dire need for a treatment option able to overcome many mechanisms of resistance in Gram-negative bacteria,” said Akira Kato, Ph.D., president and CEO at Shionogi Inc.
The increasing resistance of many infections caused by Gram-negative bacteria to existing therapies, including carbapenem-resistant Enterobacteriaceae and non-fermenting species such as P. aeruginosa, Acinetobacter baumannii (A. baumannii), and Stenotrophomonas maltophilia (S. maltophilia), poses a serious health challenge.
There is an increasing number of Gram-negative pathogens resistant to multiple antibiotics, making them difficult to treat and resulting in high mortality rates.6 In the United States, more than 2.8 million antibiotic-resistant infections occur each year, and more than 35,000 people die as a result.7 If no action is taken, antibiotic resistance is predicted to kill 10 million people every year by 2050 at a cumulative cost to global economic output of 100 trillion USD.8 The World Health Organization (WHO) and the Centers for Disease Control and Prevention have identified carbapenem-resistant strains of Enterobacteriaceae, P. aeruginosa, and A. baumannii as the top priorities in the research and development of new antibiotics.2,7
“We are proud to make FETROJA available for patients and their infectious disease care teams. Currently, FETROJA is the only available antibiotic which provides in vitro coverage against all Gram-negative pathogens considered top priority by the WHO,” said Nate McCutcheon, Chief Commercial Officer, Shionogi Inc. “At Shionogi, we are currently expanding our organization to better assist hospitals and infectious disease specialists to support this community in dire need.”
FETROJA® (cefiderocol) is a cephalosporin antibiotic with a novel mechanism for penetrating the outer cell membrane of Gram-negative pathogens by acting as a siderophore. In addition to entering cells by passive diffusion through porin channels, FETROJA binds to ferric iron and is actively transported into bacterial cells through the outer membrane via the bacterial iron transporters, which function to incorporate this essential nutrient for bacteria.9 These mechanisms allow cefiderocol to achieve high concentrations in the periplasmic space where it can bind to penicillin-binding proteins and inhibit cell wall synthesis in the bacterial cells.
FETROJA has also demonstrated in vitro activity against certain bacteria that contain very problematic resistant enzymes such as ESBLs, AmpC, serine- and metallo-carbapenemases.9 Data from multinational surveillance studies for FETROJA demonstrated potent in vitro activity against a wide spectrum of Gram-negative pathogens including carbapenem-resistant A. baumannii, P. aeruginosa, Enterobacteriaceae, and S. maltophilia.1 The clinical significance of the in vitro data is unknown. FETROJA has poor in vitro activity against Gram-positive or anaerobic bacteria.
Shionogi also submitted a marketing authorization application for cefiderocol to the European Medicines Agency and it was accepted in March 2019.
Source: Company Press Release
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.