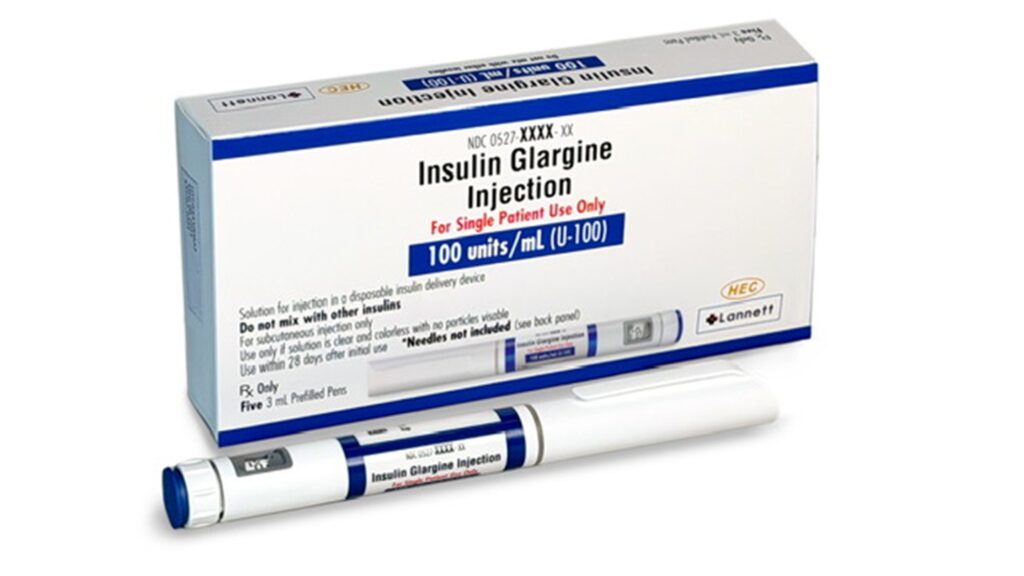
Lannett, Lanexa Biologics and Sunshine Lake secure FDA approval for Langlara
Sunshine Lake Pharma, Lannett and its wholly owned subsidiary Lanexa Biologics have received approval from the US Food and Drug Administration (FDA) for Langlara (insulin glargine-aldy), an interchangeable biosimilar of Lantus (insulin glargine).
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.